|
which is the correct, balanced equation for. The nuclear equation must contain all the reactants and products and we must ensure that the mass numbers and atomic numbers balance on either side of the equation. The universe is full of instances of nuclear fusion reactions. Write the nuclear equation for this fission reaction. Similarly, the sum of the atomic numbers of the products 46 + 52 + (4 × 0) 98 is the same as the atomic number of the parent nuclide. Examples may be given of typical reaction products, such as: U-235 + n > Ba-144 + Kr-90 + 2n + about. The fission reaction in U-235 produces fission products such as Ba, Kr, Sr, Cs, I and Xe with atomic masses distributed around 95 and 135. Thus the sum of the mass numbers of the products (118 + 132 + 4 254) equals the mass number of the reactant. However, conservation laws require the total number of nucleons and the total energy to be conserved. Nuclear fusion is a nuclear reaction that combines two or more small atoms to form a large atom. Once again, the number of nucleons is conserved. Some of them produce neutrons, called delayed neutrons, which contribute to the fission chain reaction. Nuclear fission is a nuclear reaction that splits a heavy atom into multiple smaller ones. Calculate the energies of particles produced by a fission reaction Explain the fission concept in the context of fission bombs and nuclear reactions In 1934, Enrico Fermi bombarded chemical elements with neutrons in order to create isotopes of other elements. A chain reaction can produce self-sustained fission if each fission produces enough neutrons to induce at least one more fission. A critical chain reaction can be achieved at low concentrations of U-235 if the neutrons from fission are moderated to lower their speed, since the probability for fission with slow neutrons is greater.Ī fission chain reaction produces intermediate mass fragments which are highly radioactive and produce further energy by their radioactive decay. Most nuclear power reactors use uranium-235 as the target nucleus into which a neutron is accelerated to split the. These two nuclear processes use the binding energy of the protons and neutrons in the nucleus of atoms to release an enormous amount of energy. Nuclear fission was discovered on 19 December 1938 in Berlin by German chemists Otto Hahn and. 
The fission process often produces gamma photons, and releases a very large amount of energy even by the energetic standards of radioactive decay. If the reaction will sustain itself, it is said to be "critical", and the mass of U-235 required to produced the critical condition is said to be a " critical mass". Fission and fusion are nuclear reactions that produce energy. Nuclear fission is a reaction in which the nucleus of an atom splits into two or more smaller nuclei. /GettyImages-103311355-490ad3ba66d44d40b738a0e7d468ac8a.jpg)
If an least one neutron from U-235 fission strikes another nucleus and causes it to fission, then the chain reaction will continue. If at least one neutron from each fission strikes another U-235 nucleus and initiates fission, then the chain reaction is sustained. 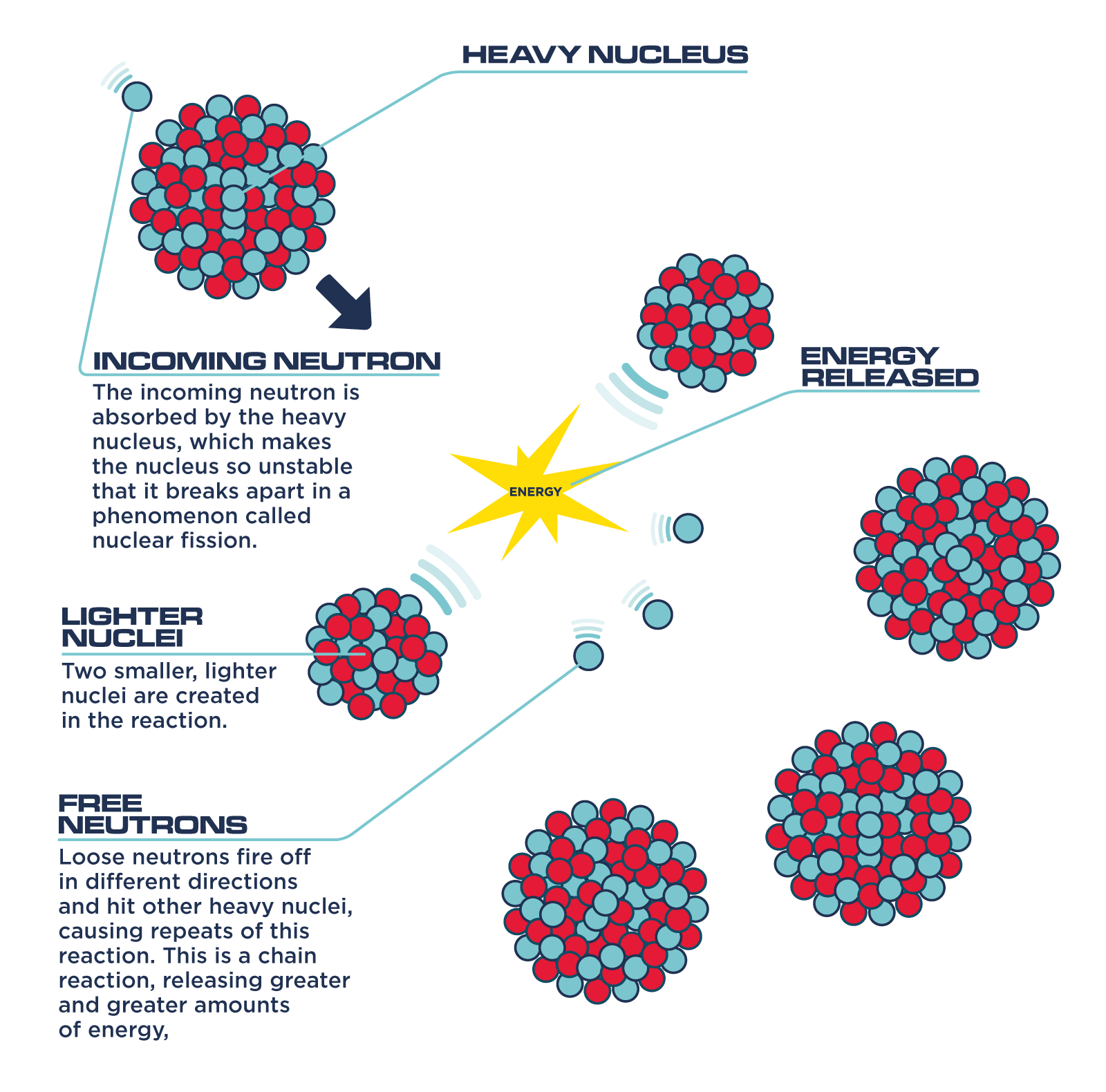 
Fission reaction does not normally occur in nature. Fusion is the fusing of two or more lighter atoms into a larger one. Uranium-235 Fission Example Initiation of this processĮnergy From Uranium Fission Form of Energy ReleasedĮnergy of decay products of fission fragments Calculating Energy from a Kilogram of Fissionable Fuel Calculate the amount of energy produced by the fission of 1.00 kg of 235U 235 U, given the average fission reaction of 235U produces 200 MeV. Fission is the splitting of a large atom into two or more smaller ones. The new generation of stars begins the nucleosynthesis process anew, with a higher percentage of heavier elements.Uranium-235 Chain Reaction Uranium-235 Fission Detailed example
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
